Introduction
One of the most prevalent and quickly spreading arbovirus infections, dengue fever poses a serious threat to public health in developing nations like India. According to the Global Burden of Dengue Fever study, symptomatic dengue diseases claim the lives of roughly 10,000 individuals annually [1]. Its endemicity affects more than 100 countries in America, Africa, South-East Asia, Eastern Mediterranean, and Western Pacific [2]. Dengue infection is produced by four different viral serotypes, and its clinical symptoms are the outcome of intricate interactions between the virus and host vector that are impacted by climate [3].
The viraemic phase, the host's immunological response, and haematological trends in peripheral blood are all tightly linked to the dynamic course of dengue fever. Plasma leakage that causes shock and thrombopathy that causes bleeding are the two main pathophysiological features of dengue [4]. Although there isn't a specific therapy for dengue fever, early therapeutic treatments have been demonstrated to reduce death rates to less than 1%. In order to facilitate the reabsorption of extravasated fluid, interventions include de-escalating fluid therapy toward the end of the plasma leakage phase and increasing fluid therapy during the active period [5].
The most crucial indicators to monitor during the acute and progressive stages of the disease are rising haematocrit and falling platelet count. It is crucial to comprehend and choose the right early markers of disease progression [6]. An automated measurement of reticulated platelets (newly released platelets that mature in 24 to 48 hours) in peripheral blood is called immature platelet fraction (IPF) [7].
IPF % is a promising method predicting the platelet count trajectory in patients with dengue since the presence of reticulated platelets is an indirect indicator of bone marrow activity prior to the rise of platelets in the blood. An increase in the IPF percentage indicates that the platelet count will rise within the following 24 to 48 hours [8]. In order to prevent needless platelet transfusions, which can result in consequences such alloimmunization, immunosuppression, infectious disease transmission, and graft vs. host illness, our goal in this study is to quantify the IPF in pediatric patients in order to predict platelet recovery [9]. Studies evaluating the effectiveness of Immature Platelet Fraction and platelet recovery in children under the age of twelve who have dengue fever are extremely rare. Thus, we conducted the present study to determine the relationship between IPF and platelet count and evaluated sensitivity and specificity of IPF and predict the cut-off point of IPF with platelet count recovery in dengue affected children age less than 12 years.
Material and Method
The present prospective observational cohort study was conducted in the Department of Paediatrics from May 2022 till June 2023, on 100 children aged <12yrs of age, who were IgM Dengue positive or NS1antigen positive or both, with platelet count less than 1.5 lakh. Children having platelet count less than 1.5 lakhs due to other reasons such as Immune thrombocytopenia, Leukaemia, aplastic anaemia; previously platelet transfused cases; and those requiring transfusion were excluded from the study. Taking sensitivity and specificity of IPF from study by Agarwal M et al.,[10], the minimum required sample size with desired precision of 15%, 80% power of study and 5% level of significance was calculated to be 84. To reduce the margin of error, total sample size was taken as 100.
Parents/ guardians of all included cases were detailed about the study and a written informed consent was taken. 3ml of blood sample was taken on the day of admission; at 3rd, 5th, 7th day and at day of discharge for assessing immature platelet fraction through mindray automated analyser. Immature platelet fraction and platelet count was assessed in enrolled patients. For co-relation IPF% with recovery of platelet count, criteria for recovery was taken as increased in platelet count by 20,000 or above. Sensitivity and specificity analysis was carried out for observation of platelet count below 1.5 lakh for all patient on day of admission 3,5,7 and day of discharge; and predict the cut off of IPF on corresponding days and recovery in platelet count by 20,000 or more in subsequent 48hrs. Data collected was analyzed using IBM SPSS (SPSS Inc., IBM Corporation, NY, USA) Statistics Version 25 for Windows software program at level of significance set at p<0.05.
Results
In our study, dengue illness was most commonly observed in 60-119 months old children, followed by 120-144 months old (20%), and 0-59 months old (16%). Mean age of children suffering from dengue was found to be 8.11±2.51yrs. Dengue illness was more common in males (58%) than females (42%). In our study, Mean platelet count increased from 57,713.38 /μl at day 1; to 77495.68 /μl at day 3; 111649.09/μl at 5th day and 1,72,612.58 /μl at discharge. Mean changes was 19782.3; 34153.41 and 60963.49 on every follow up. Mean IPF was 5.71% at day 1; 6.65% at day 3; 11.86% at 5 days and 5.34% at discharge. Mean changes average was 0.94%, 5.21% and 6.5% on every follow up.
In our study Platelet was significantly increased after day 3, day 5 and at the time of discharge before day 7. IPF was significantly increased only at the time of day 3 and day 5. IPF was negative significant correlation at discharge before day 7 (Table 1). Platelet count was significantly (p<0.05) correlated with IPF at day 1 and 5 (Table 2).
In our study at day 3, sensitivity was 80.56%, specificity was 40.71%, PPV was 48.53%, NPV was 89.31% and accuracy was 59.27%. At day 5, sensitivity was 75.69%, specificity was 80.53%, PPV was 76.23%, NPV was 62.8% and accuracy was 83.61%. At day 7, sensitivity was 85.36%, specificity was 95.36%, PPV was 93.47%, NPV was 52.61% and accuracy was 83.97% (Table 3). Area under curve was assessed for both PLT and IPF (Table no. 4); and ROC curves were created (Figure 1 & 2).
| Platelet wise distribution | DAY 1 | DAY 3 | DAY 5 | At the time of discharge before day 7 | |
| DAY 1 | Pearson Correlation | 1 | .405** | .590** | .543** |
| P value | .000 (S) | .000 (S) | .000 (S) | ||
| DAY 3 | Pearson Correlation | .405** | 1 | .326** | .247* |
| P value | .000 (S) | .001 (S) | .01 (S) | ||
| DAY 5 | Pearson Correlation | .590** | .326** | 1 | .591** |
| P value | .000 (S) | .001 (S) | .000 (S) | ||
| At the time of discharge before day 7 | Pearson Correlation | .543** | .247* | .591** | 1 |
| P value | .000 (S) | .01 (S) | .000 (S) | ||
| IPF wise distribution | |||||
| DAY 1 | Pearson Correlation | 1 | .149 | .179 | -.219* |
| P value | .02 (S) | .03 (S) | .02 (S) | ||
| DAY 3 | Pearson Correlation | .149 | 1 | .461** | .167 |
| P value | .02 (S) | .000 | .098 | ||
| DAY 5 | Pearson Correlation | .179 | .461** | 1 | .223* |
| P value | .03 (S) | .000 | .02 (S) | ||
| At the time of discharge before day 7 | Pearson Correlation | -.219* | .167 | .223* | 1 |
| P value | .02 (S) | .098 | .026 | ||
| N | 100 | 100 | 100 | 100 |
| PLT | IPF | |
| Day 1 | Pearson Correlation | .264** |
| P value | .008 | |
| Day 3 | Pearson Correlation | -.103 |
| P value | .306 | |
| Day 5 | Pearson Correlation | -.331** |
| P value | .001 | |
| Day 7 | Pearson Correlation | -.040 |
| P value | .696 |
| Day 3 | Day 5 | Day 7 | |
| Sensitivity | 80.56 | 75.69 | 85.36 |
| Specificity | 40.71 | 80.53 | 95.36 |
| PPV | 48.53 | 76.23 | 93.47 |
| NPV | 89.31 | 62.8 | 52.61 |
| Accuracy | 59.27 | 83.61 | 83.97 |
| Area Under Curve | p-value | Asympto95% Confidence Interval | ||
| Lower Bound | Upper Bound | |||
| IPF DAY 3 | .429 | .01 (S) | .312 | .546 |
| IPF DAY 5 | .595 | .01 (S) | .460 | .731 |
| IPF DAY 7 | .673 | .007 (S) | .558 | .788 |
| PLT DAY 3 | .826 | .01 (S) | .662 | .991 |
| PLT DAY 5 | .866 | .006 (S) | .782 | .951 |
| PLT DAY 7 | .486 | .01 (S) | .299 | .674 |
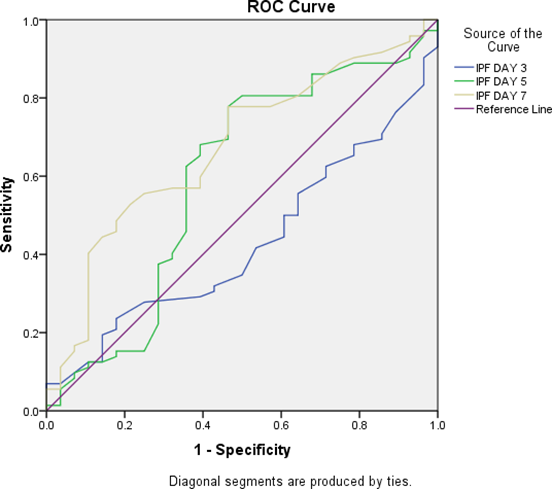
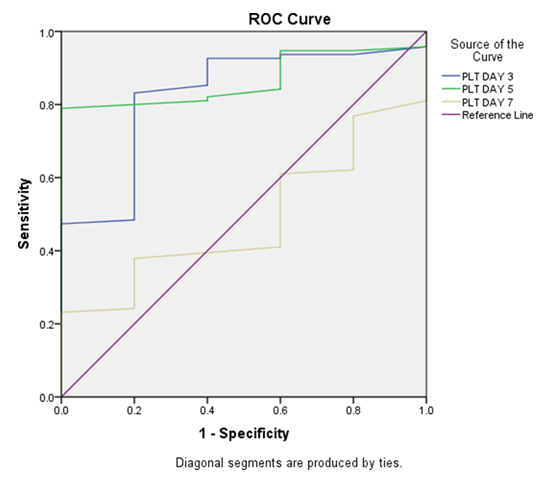
Discussion
IPF has developed into a trustworthy future reference for choices pertaining to platelet transfusions in recent years. Rise in base IPF levels are directly correlated with increases in platelet count, according to earlier research. Therefore, measuring IPF should be a standard procedure for tracking thrombocytopenia in dengue patients. In order to prevent needless blood transfusions, the treating physician can use IPF to forecast the recovery of platelets in dengue patients [11].
In the present study, the most common age group of children affected with dengue was 60-119mths; which is consistent with the study by Chairulfatah A et al.,[12] We observed male predominance in our study, with male to female ratio being 1.38:1. In accordance with our research, studies by Paul M et al.,[13], Puspita R et al.,[14], and Chatterjee AB et al.,[15] also found male predominance (68%, 60% and 56% respectively). In contrast to our study, Ahmed J et al.,[16] observed female predominance.
Immune-pathology is one of the main causes of dengue. Changes in the host's physiology during the dengue virus eradication process can be beneficial after a time of acute illness, but in situations where the host's physiology is not resilient enough to tolerate the shift (such as in children or older adults), it may cause irreversible harm [17]. Sara et al.,[18] reported IPF as an indicator of thrombopoietic state whereas Briggs et al.,[19] concluded that an increase in IPF% preceded thrombocytopenia recovery in chemotherapy and transplant patients.
In our present study, platelet count was significantly increased from day 3, till the time of discharge, whereas IPF was significantly increased only at day 3 and 5. IPF showed a negative but significant correlation at the time of discharge. Similar findings were observed in studies by Agarwal M et al.,[10], and Dadu T et al.,[20]; whereas Ezhilarasu R et al.,[21] observed an insignificant correlation between changes in platelet count and immature platelet fraction.
In our study it is found that a strong positive correlation between IPF and platelet count recovery. Therefore, IPF might be regularly monitored to evaluate the thrombopoiesis in dengue-affected children. In particular, platelet recovery from the nadir of thrombocytopenia on days 4-6 of illness confirmed the results of earlier studies. IPF% increased for more than three days before platelet recovery, indicating that IPF% is a promising tool that can predict the platelet count trajectory in cases suffering with dengue [22-24]. Hence IPF shows great promise of becoming a reliable future guide for decisions concerning platelet transfusions.
The reference range for IPF utilized in our study was 5-11%, minimum IPF measured was 5.7% and maximum 11.8%, which is similar to study by Lavanya N et al.,[25] with mean IPF being 11.67% (2% to 26.9%). In another studies by Agarwal M et al.,[10] Briggs et al.,[19] and Dadu T et al.,[20], the ranges were 0.7-4.3%; 1.1-6.1% and 0.9-5.4% respectively. In our study, values of sensitivity (80.56% to 85.36%) and specificity (40.71% to 95.36%) increased from day 3 to day 7. In study by Agarwal M et al.,[10] at day 3, sensitivity and specificity was 85.37% and 38.38%. At day 7, sensitivity was found to be 77.42%, and specificity was 100%. Ong -Misa MM et al.,[26] found IPF value of 6.6%, with specificity of 70% and sensitivity of 45%. IPF cut-off value of 6.25% (specificity and sensitivity of 63% and 77% respectively) was determined by Suman et al.,[27] which was statistically significant in predicting platelet recovery within 48 hours.
Our study has few limitations: We have utilized the obtained values from the prior study because there is a dearth of knowledge and data to serve as a reference for platelet indices in children. The study was conducted at a single location, which is representative of dengue patients in other locations and nations, and the sample size was somewhat small. As a result, IPF has a strong chance of developing into a trustworthy future reference for choices pertaining to platelet transfusions. Increases in basal IPF levels and concomitant rise in platelet count seem to be directly correlated. In dengue patients, there seems to be a 24-48-hour lag between elevated IPF levels and matching increases in platelet count. In order to assess and track thrombocytopenia in dengue fever patients, IPF measurement should be regarded as standard procedure.
Conclusion
IPF can be used to assess a dengue patient's platelet recovery. In the future, IPF will be a trustworthy resource for making judgments about platelet transfusions. Increases in basal IPF levels seem to be directly correlated with increases in platelet count, allowing for the postponement of prophylactic platelet transfusion and its risks. Therefore, measuring IPF should be regarded as standard procedure for tracking and assessing thrombocytopenia in dengue patients.
Declaration
Ethical Statements
The ethical committee approved the study. [IRB Approval letter number and date (IEC-DDU/upn75/2022-05-05/5/v1) (10-05-2022)] Informed written consent was obtained from the parents/guardians of the children and the ascent was obtained from children above ten years of age.
Conflicts of Interest
The authors have nothing to disclose.
Acknowledgement
We thank all the residents and staff who were involved in the care of this patient.
Financial Support and Sponsorship
Nil.